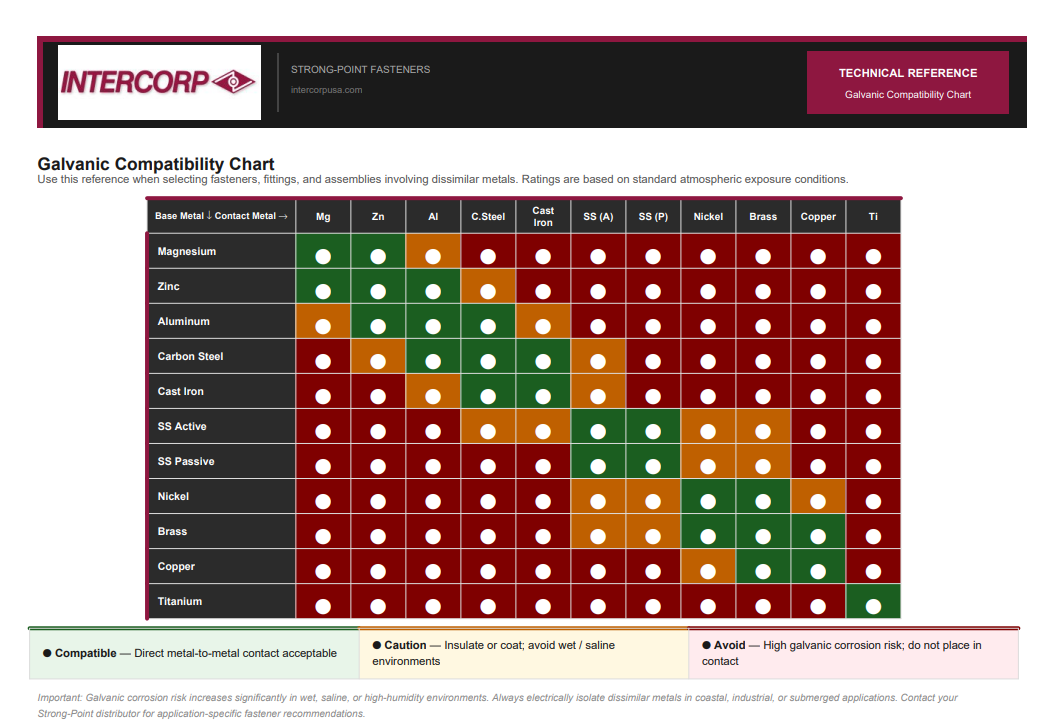
Galvanic Corrosion Chart: Check Metal Compatibility Instantly
Prevent costly failures caused by dissimilar metals. Use this galvanic corrosion chart to quickly assess compatibility and reduce corrosion risk in your application.
Galvanic Compatibility Checker
Select a base metal and a contact metal to see estimated galvanic corrosion risk and the recommended next step. Green/Compatible assumes dry, controlled environments.
- Coastal and salt-heavy environments accelerate galvanic action.
- Standing water, condensation, and trapped moisture increase failure risk.
- Surface coatings, finish condition, and fastener design can materially change real-world performance.
Intercorp can review your metal pairing and recommend a more durable solution.
What Is Galvanic Corrosion?
Galvanic corrosion occurs when two dissimilar metals come into electrical contact in the presence of an electrolyte (like moisture or saltwater). One metal becomes the anode and corrodes faster, while the other is protected.
This reaction is driven by differences in the galvanic series of metals, which ranks materials by their electrochemical potential.
Galvanic Corrosion Metals: Why Compatibility Matters
Not all metals behave the same when paired together. The further apart two metals are in the galvanic series chart, the greater the risk of corrosion.
Key Factors That Increase Risk:
- Moisture or humidity
- Salt exposure (coastal environments)
- Chemical exposure
- Poor drainage or trapped water
Even metals with moderate compatibility can fail prematurely in harsh environments.
How to Read a Galvanic Corrosion Chart
Using a galvanic reaction chart is straightforward:
- Identify the base (corroding) metal
- Find the contact metal
- Locate where they intersect
- Review the corrosion risk level:
- Low Risk → Generally safe
- Moderate Risk → Use protection
- High Risk → Avoid pairing
Galvanic Series Chart Explained
A galvanic series chart ranks metals from most anodic (more likely to corrode) to most cathodic (less likely to corrode).
Simplified Order (Most to Least Reactive):
- Magnesium
- Zinc
- Aluminum
- Carbon Steel
- Stainless Steel
- Nickel
- Copper
- Titanium
The greater the distance between two metals in this series, the higher the risk of dissimilar metals corrosion.
Dissimilar Metals Corrosion: Real-World Impact
When incompatible metals are used together, it can lead to:
- Premature fastener failure
- Structural weakening
- Increased maintenance costs
- Safety risks in load-bearing systems
This is especially critical in:
- Construction
- Roofing systems
- Marine environments
- Industrial applications
Galvanic Corrosion Prevention Strategies
Preventing corrosion is not just about choosing the right metals, it’s about controlling the environment and contact points.
Proven Methods:
1. Use Compatible Metals
Select metals close together in the galvanic corrosion chart whenever possible.
2. Apply Protective Coatings
Barrier coatings prevent direct metal-to-metal contact and reduce corrosion risk.
3. Use Isolation Materials
Non-conductive washers, sleeves, or gaskets can break the electrical connection.
4. Control the Environment
Reduce moisture exposure and improve drainage to limit electrolytes.
5. Choose the Right Fastener System
Coated or corrosion-resistant fasteners can significantly extend lifespan.
When to Be Most Concerned
You should pay extra attention to galvanic corrosion when:
- Metals are exposed to outdoor environments
- There is frequent moisture or condensation
- Systems are near saltwater or chemicals
- Long-term durability is critical
Need Help Preventing Galvanic Corrosion?
Choosing the right materials and coatings can prevent expensive failures and extend the life of your project.
FAQ: Galvanic Corrosion
What metals should not be used together?
Metals far apart in the galvanic series—such as aluminum and copper—should generally be avoided without protection.
Does stainless steel cause corrosion with other metals?
Yes. Stainless steel can accelerate corrosion when paired with more anodic metals like aluminum or carbon steel.
How do you stop galvanic corrosion?
Use coatings, isolate metals, and select compatible materials based on a galvanic corrosion chart.